Bitte besuchen Sie die deutsche Seite der AG Leprivier hier.
“Stress responses and Cancer” lab
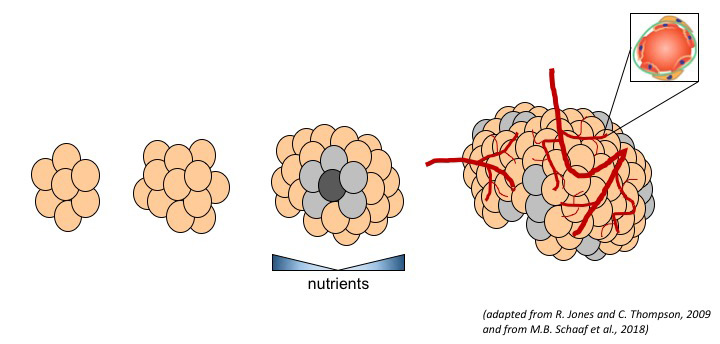
Tumors develop within very hostile microenvironments, which are characterized by various stress conditions, including nutrient deprivation, hypoxia, oxidative stress, or acidosis. In particular, tumors have limited access to nutrients as a result of defects in the tumor vasculature. While such conditions initially restrict tumor progression, certain tumor cells can adapt, leading to the emergence of more aggressive cancer cell clones. It is critical to delineate the mechanisms underlying tumor adaptation to nutrient stress. This has the power to uncover novel therapeutic targets with high selectivity, especially since normal tissues are not experiencing such a severe nutrient stress.
We are interested to identify and characterize novel drivers of tumor adaptation to nutrient stress which are relevant to brain cancers, including medulloblastoma and glioblastoma. Our works focus on the role of cell signaling components, regulators of mRNA translation, as well as long non-coding RNAs, a new class of genes. Specifically, we are investigating the molecular mechanisms involved in stress protection, and are elucidating their contribution to tumor development, progression and relapse. This is complemented by biomarker development, to focus on molecules with particular clinical significance, and design of therapeutic strategies for brain cancer patients.
We are using a wide range of molecular biology and cellular biology techniques to functionally investigate the role of protein and RNA candidates and model brain cancer.

Applications from highly motivated students and post docs are welcome.
Selected publications:
Leprivier G, Remke M, Rotblat B, Dubuc A, Mateo AR, Kool M, Agnihotri S, El-Naggar A, Yu B, Somasekharan SP, Faubert B, Bridon G, Tognon CE, Mathers J, Thomas R, Li A, Barokas A, Kwok B, Bowden M, Smith S, Wu X, Korshunov A, Hielscher T, Northcott PA, Galpin JD, Ahern CA, Wang Y, McCabe MG, Collins VP, Jones RG, Pollak M, Delattre O, Gleave ME, Jan E, Pfister SM, Proud CG, Derry WB, Taylor MD, Sorensen PH. The eEF2 kinase confers resistance to nutrient deprivation by blocking translation elongation. Cell, 2013 May 23;153(5):1064-79. PubMed
Lim JKM, Delaidelli A, Minaker SW, Zhang HF, Colovic M, Yang H, Negri GL, von Karstedt S, Lockwood WW, Schaffer P, Leprivier G*, Sorensen PH*. Cystine/glutamate antiporter xCT (SLC7A11) facilitates oncogenic RAS transformation by preserving intracellular redox balance. Proc Natl Acad Sci U S A, 2019 May 7;116(19):9433-9442. *Co-senior authorship. PubMed
Follow us on Twitter: @AGLeprivier.