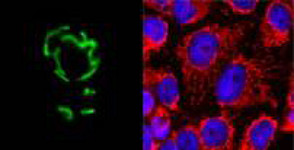
Mitochondrial dynamics and quality control of mitochondria
Our group aims to elucidate the link between mitochondrial dynamics and degradation of damaged mitochondria by mitophagy and to decipher the mechanisms determining mitochondrial ultrastructure. The mitochondrial network in cells is highly dynamic. Mitochondrial fusion and fission as well as redistribution and degradation of mitochondria do occur constantly. Fusion and fission are predicted to be essential for quality control of mitochondria as in many diseases the balance between fusion and fission is affected. Furthermore, dysfunctional mitochondria are unable to fuse to the intact mitochondrial network and it was proposed that this is a prerequisite for distinguishing functional from dysfunctional mitochondria. We study the process of selective degradation of mitochondria as dysfunctional mitochondria can be degraded via mitophagy and an impaired degradation and the consequential accumulation of dysfunctional mitochondria seem to play an important role in the aging process and the pathogenesis of numerous human disorders.
Another focus of our research is to understand the molecular mechanisms that shape the inner mitochondrial membrane, particularly, cristae and crista junctions and their importance in cellular viability.
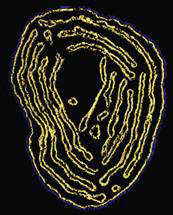
Fig. 2: Cristae morphology in a yeast strain lacking Fcj1. A surface rendered view of an EM tomogram of a mitochondrium is shown (from Rabl et al. 2009). The inner membrane (yellow) shows cristae membranes arranged in parallel concentric stacks lacking crista junctions.