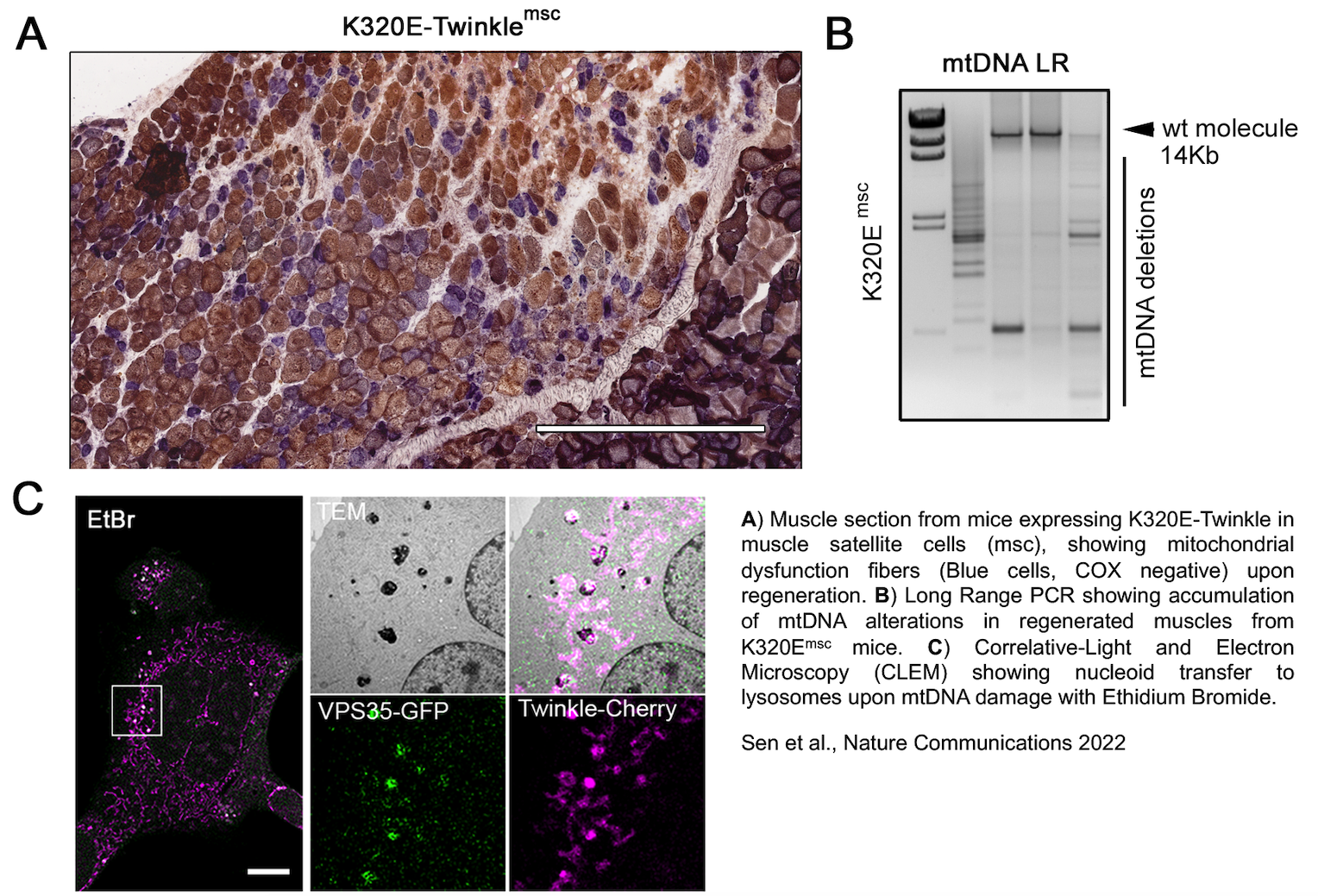
mtDNA, Quality Control and Aging
Accumulation of mtDNA mutations is a natural process occurring in all long-living organisms. The multicopy nature of the mitochondrial genome, varying from hundreds to thousands of copies per cell, permits that mitochondrial dysfunction only strikes when the number of mtDNA mutations surpasses a specific threshold. In this scenario, quality control mechanisms such mitophagy coordinate to tackle cellular burden, removing damaged mitochondria, and compensating mtDNA mutation load. However, as other cellular surveillant pathways, mitophagy is compromised with aging, leading to a gradual accumulation of mitochondrial and mtDNA damage.
The mitochondrial genome (mtDNA), owing to its resemblance to a bacterial genome, is immunostimulatory. Upon mitochondrial dysfunction, mtDNA leaks to the cytosol, supporting low-level inflammatory state which gradually increases with age and contributes to cellular burden. Our lab aims to investigate the relationship between the natural accumulation of mitochondrial damage, quality control of the mitochondrial genome, and activation of the innate immune response, as mechanisms supporting the development of age-related disease.
For our research we use the state-of-the-art for protein identification, light and electron microscopy, cellular and mouse models. We have developed several tools to damage in vitro the mitochondrial genome. We combine these tools with proximity proteomics to determine specific pathways for mtDNA turnover. In addition, we use a transgenic mouse model with progressive accumulation of mtDNA alterations, to study the contribution of mtDNA damage to the development of age-related movement disorders. For that, we target specifically motor neurons in the peripheral nervous system and Purkinje Cells in the cerebellum.